Pharmaceuticals in West Africa: Compliance, Demand & Distribution

Africa's pharmaceutical sector is undergoing a fundamental transformation. Rising populations, expanding public health infrastructure, growing private healthcare provision, and increasing government investment in medicines access are collectively creating one of the most significant and sustained pharmaceutical import opportunities in the world. For exporters who can navigate the compliance requirements and distribution dynamics of this market, the long-term commercial potential is substantial.
This is not a simple market. Pharmaceutical trade in Africa involves complex regulatory requirements, critical compliance obligations, and distribution challenges that differ meaningfully from other product categories. But complexity creates barriers, and barriers create value for exporters who invest in understanding how to clear them properly.
The Scale of the Opportunity
Sub-Saharan Africa imports between 70% and 90% of its total pharmaceutical requirements. This figure reflects both the current limitation of local production capacity and the scale of the gap that foreign exporters can fill. While African pharmaceutical manufacturing is growing, encouraged by government policy, international development investment, and post-COVID supply chain consciousness, the gap between local production and consumer demand will remain substantial for at least the next decade.
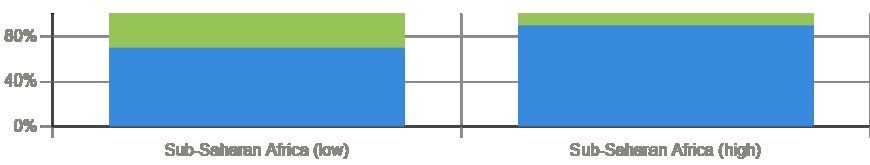
Africa's population is projected to double by 2050, with the sharpest growth concentrated in countries that currently have very limited pharmaceutical manufacturing infrastructure. Urban populations, typically the primary market for imported pharmaceuticals, are growing especially fast. Combined with rising household incomes and government commitments to expanding healthcare access, this demographic trajectory creates durable, growing demand.
The market is also diversifying. Beyond the traditional focus on essential medicines and generic pharmaceuticals, demand is growing for over-the-counter health products, medical devices and consumables, nutritional supplements, and personal care products with health positioning. Each of these sub-categories presents distinct opportunities for exporters with relevant product portfolios.
Nigeria is Africa's largest pharmaceutical market by volume. Its population of over 220 million people, combined with a rapidly expanding private healthcare sector and significant unmet demand for quality medicines, creates a substantial import opportunity. The regulatory authority, NAFDAC (National Agency for Food and Drug Administration and Control), has a comprehensive product registration framework that requires careful navigation, but registration opens access to the continent's most commercially significant market.
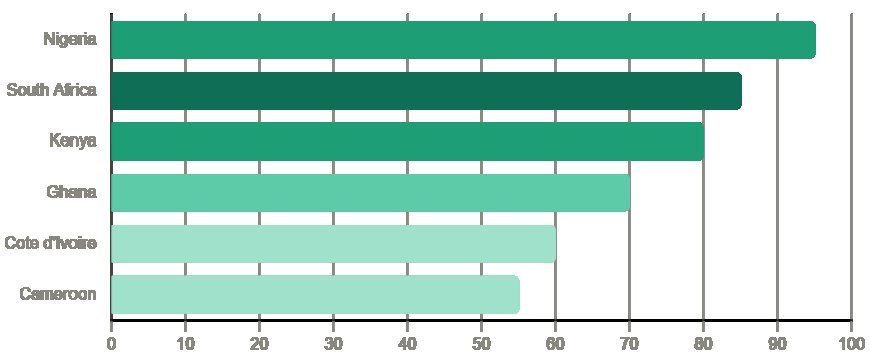
Kenya is East Africa's pharmaceutical hub. Nairobi serves not only the Kenyan market but, through its well-developed distribution networks, supplies importers and distributors across Uganda, Rwanda, Tanzania, Ethiopia, and the broader East African Community. Kenya's Pharmacy and Poisons Board is one of the more efficient regulatory bodies on the continent, and its registration processes, while thorough, are generally more predictable than some regional counterparts.
Ghana has built a reputation as one of West Africa's most reliable pharmaceutical import markets. The Food and Drugs Authority (FDA Ghana) operates a transparent registration process, and Ghana's stable political environment and English-speaking business culture make it a natural first entry point for pharmaceutical exporters seeking West African market access. Accra's pharmaceutical distribution infrastructure serves both domestic retailers and cross-border trade into Francophone neighbours.
Cameroon and Côte d'Ivoire are the primary pharmaceutical import markets in Central and Francophone West Africa, respectively. Both markets require French-language documentation for most regulatory submissions. Importers in these markets often combine pharmaceutical distribution with broader healthcare product portfolios, which creates cross-selling opportunities for exporters who can serve multiple categories.
South Africa, while technically Southern rather than Sub-Saharan Africa in some classifications, merits mention as the continent's most sophisticated pharmaceutical market. Its regulatory body (SAHPRA) operates to standards comparable to European regulators. South Africa serves as a regional headquarters for many international pharmaceutical companies and as a distribution gateway for the broader Southern African Development Community (SADC) region.
Compliance: What You Cannot Afford to Get Wrong
Pharmaceutical export compliance in Africa is non-negotiable. The consequences of non-compliance include product seizure, import bans, regulatory sanctions against your local importer, and potentially serious harm to end users. This is a category where every detail of the compliance framework must be understood and met.
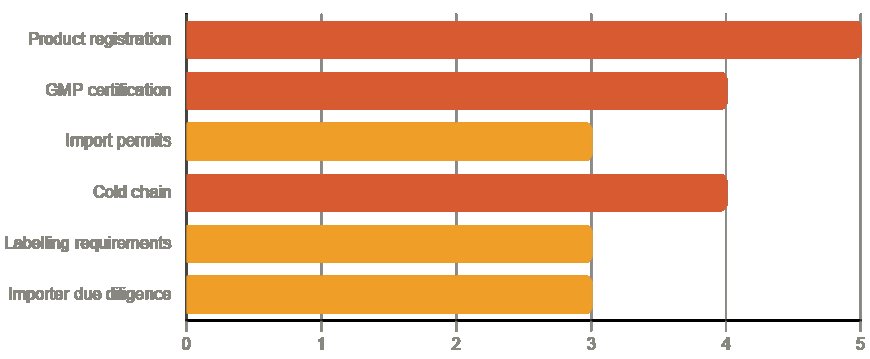
Product registration is the foundational requirement. Every pharmaceutical product must be registered with the national medicines regulatory authority of the target market before it can be legally imported and sold. Registration processes vary by country in terms of timeline, documentation requirements, and fees, but typically require: a dossier covering product composition, manufacturing process, quality control standards, and clinical evidence; a Certificate of Pharmaceutical Product (CPP) from the country of manufacture; and evidence of Good Manufacturing Practice (GMP) certification for the manufacturing facility.
WHO GMP certification, or equivalent national GMP certification from a recognised regulatory authority, is required by virtually all African medicines regulators. This certifies that the manufacturing facility meets internationally accepted standards for pharmaceutical production quality and consistency. Without it, product registration applications will not be accepted.
Import permits are typically required in addition to product registration. Most African countries require importers to obtain specific import permits for each pharmaceutical shipment, presenting the registered product and confirming the approved source and quantity. Your importer should be managing this process, but understanding the requirement helps you plan shipment timing appropriately.
Cold chain requirements apply to a significant and growing portion of the pharmaceutical export market. Vaccines, biologics, insulin, and many speciality medicines require temperature-controlled shipping and storage throughout the supply chain. Not all African logistics providers have cold chain capability to the required standard, so confirming cold chain infrastructure end-to-end before committing to temperature-sensitive product categories is essential.
Labelling requirements are exacting and vary by market. Most African regulators require product labels in specific languages (English in Anglophone markets, French in Francophone markets), with mandatory information including batch number, manufacturing and expiry dates, storage conditions, active ingredient concentrations, and the name and address of the registered importer. Labelling non-compliance is one of the most common reasons for pharmaceutical shipments being held at customs.
Distribution Dynamics
Pharmaceutical distribution in Africa is structured around a relatively small number of established importers who serve as the interface between foreign manufacturers and the fragmented downstream retail and institutional market. Understanding this structure is important for calibrating your commercial strategy.
At the top of the distribution chain are national importers and distributors, companies with the regulatory relationships, warehousing capacity, cold chain infrastructure (where relevant), and financial strength to import at scale and distribute nationally. These are the primary buyer relationships that foreign pharmaceutical exporters should target. In each major market, there are typically ten to twenty companies at this level; identifying and building relationships with the most reputable and financially stable among them is the key commercial challenge.
Below the national importer tier, distribution flows through regional wholesalers, institutional procurement (public hospitals, health ministries, and NGO procurement programmes), and retail pharmacies. Foreign exporters typically do not need to engage directly at these levels; that is the function of the national importer, but understanding the downstream structure helps you assess the quality and reach of your importer partner.
Institutional procurement channels are particularly important for certain product categories. Governments, international health organisations (WHO, UNICEF, Médecins Sans Frontières), and large NGOs purchase pharmaceutical products through formal procurement processes that can represent significant volumes. Qualifying for these procurement channels requires specific documentation and regulatory standing, but the volumes and payment reliability can be exceptional.
Choosing the Right Importer Partner
In the pharmaceutical trade, the quality of your importer partner is even more critical than in other categories. A weak or poorly regulated importer creates compliance risk for your brand and regulatory risk for both parties. Due diligence on pharmaceutical importers should be correspondingly thorough.
Verify that your importer holds the necessary regulatory licences, specifically, a valid pharmaceutical import licence from the national regulatory authority. Confirm that they have appropriate storage facilities, including cold chain capability where your products require it. Check their track record with other foreign suppliers and, where possible, speak directly with those suppliers about their experience.
ExporterIQ's verified pharmaceutical importer profiles across Nigeria, Kenya, Ghana, Cameroon, and Cote d'Ivoire have been through our standard verification process, covering business registration, sector validation, and trade role confirmation. For a category with compliance stakes as high as pharmaceuticals, starting with verified contacts is not just efficient, it is prudent risk management.
Africa's pharmaceutical market is large, growing, and structurally dependent on imports for the foreseeable future. For exporters with compliant, quality-assured products and the patience to invest in proper registration and importer relationship development, it represents one of the most rewarding long-term opportunities in global pharmaceutical trade. The compliance complexity that deters less committed exporters is precisely what creates sustainable competitive advantage for those who navigate it successfully.














Comments (0)